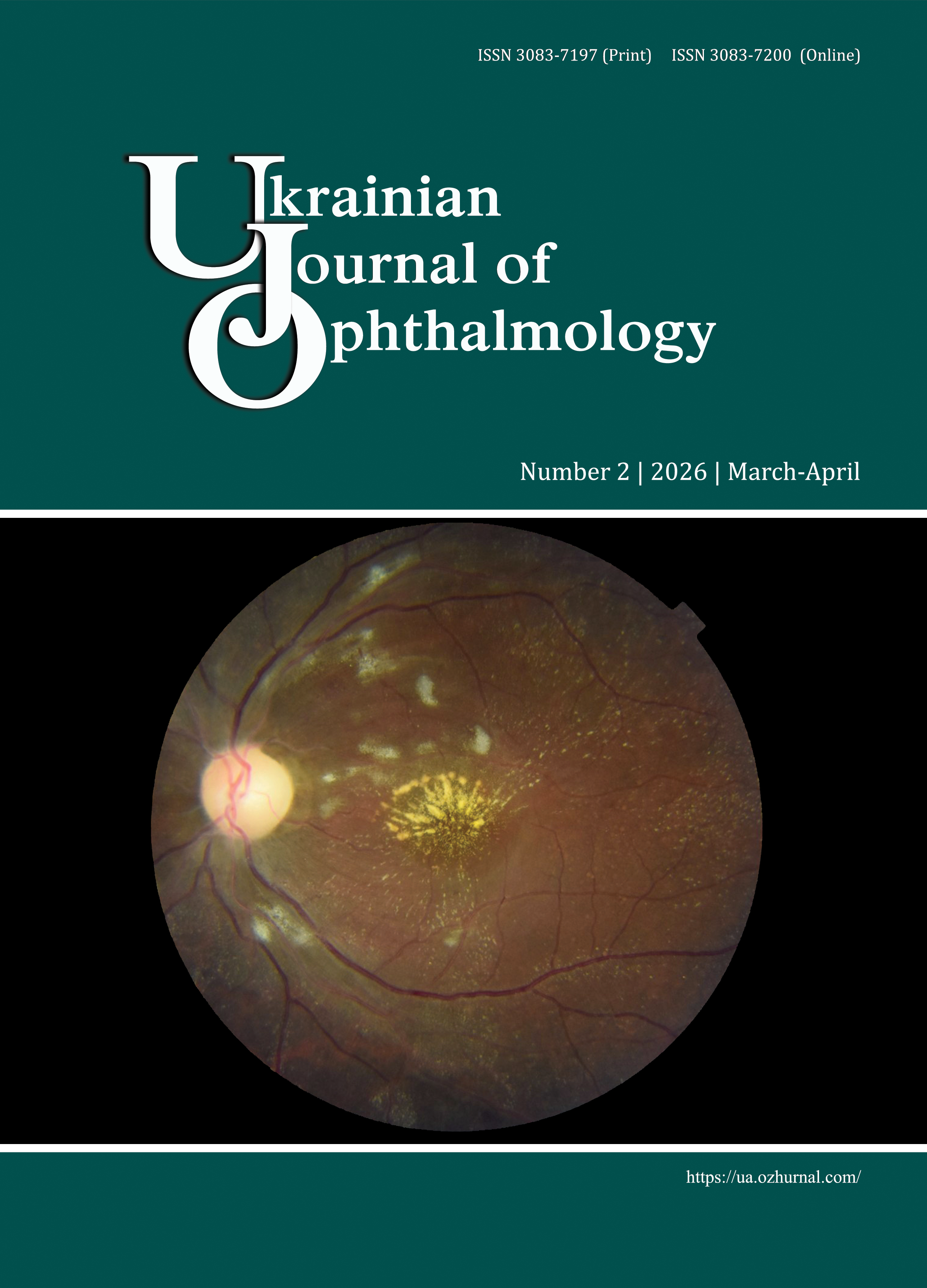
Sustained disease control with aflibercept 8 mg: A turning point in the management of exudative retinal disease
Expert consenus
DOI:
https://doi.org/10.31288/Ukr.j.ophthalmol.202627176Abstract
Leading Ukrainian retinal disease specialists participated in the Advisory Board meeting held in Kyiv in late September 2025. The meeting aimed to determine the optimal approaches to using aflibercept 8 mg in Ukraine for sustained control of, and best possible treatment outcomes for patients with, exudative retinal disease. This paper contains all the key recommendations for which all the participants achieved consensus after discussion.
References
Schubert W, Terjung C, Rafique A, Romano C, Ellinger P, Rittenhouse KD. Evaluation of Molecular Properties versus In Vivo Performance of Aflibercept, Brolucizumab, and Ranibizumab in a Retinal Vascular Hyperpermeability Model. Transl Vis Sci Technol. 2022;11(10):36. https://doi.org/10.1167/tvst.11.10.36
Regula JT, Lundhvon Leithner P, Foxton R, et al. Targeting key angiogenic pathways with a bispecific CrossMAb optimized for neovascular eye diseases. EMBO Mol Med. 2016;8(11):1265-1288. Published 2016 Nov 2. https://doi.org/10.15252/emmm.201505889
Kanda A, Noda K, Saito W, Ishida S. Aflibercept Traps Galectin-1, an Angiogenic Factor Associated with Diabetic Retinopathy. SciRep. 2015;5:17946. Published 2015 Dec 9. https://doi.org/10.1038/srep17946
Kim HM, Woo SJ. Immunogenicity and Potential for Intraocular Inflammation of Intravitreal Anti-VEGF Drugs. Curr Ther Res Clin Exp. 2024;100:100742. Published 2024 Mar 14. https://doi.org/10.1016/j.curtheres.2024.100742
Sivaprasad S, Ghanchi F, Kelly SP, et al. Evaluation of standard-of-care intravitreal aflibercept treatment practices in patients with diabetic macular oedema in the UK: DRAKO study outcomes. Eye (Lond). 2023;37(12):2527-2534.https://doi.org/10.1038/s41433-022-02367-x
Bayer AG. Eylea™ 8 mg with extended 6-month treatment interval approved in the EU. Available at: https://www.bayer.com/media/en-us/eylea-8-mg-with-extended-6-month-treatment-interval-approved-in-the-eu/. Accessed: October 2025.
Korobelnik JF, Lanzetta P, Leal S, et al. Intravitreal Aflibercept 8 mg in Neovascular Age-Related Macular Degeneration: Ninety-Six-Week Results from the Randomized Phase 3 PULSAR Trial. Ophthalmology. 2026 Jan;133(1):39-50.https://doi.org/10.1016/j.ophtha.2025.08.022
Clark WL. Three-year Outcomes of Aflibercept 8mg in Diabetic Macular Edema: Safety and Efficacy Results From the PHOTON Extension Study. Angiogenesis 2025. 8 February 2025. Virtual.
Garweg JG, Do DV, Korobelnik JF, et al. A pooled analysis of the CANDELA, PHOTON, and PULSAR trials through 96 weeks: Comparably low intraocular inflammation (IOI)-related events with aflibercept 8 mg and 2 mg. In: Proceedings of the 2025 ARVO Annual Meeting; Salt Lake City, UT, USA. 4-8 May 2025.
Konidaris V, Lange C, Munk M, et al. SPECTRUM: early clinical experience from the first global real-world study of aflibercept 8 mg in patients with treatment-naïve neovascular age-related macular degeneration*. In: Proceedings of the 2025 ARVO Annual Meeting; Salt Lake City, UT, USA. 4-8 May 2025.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Андрій Король, Ірина Безкоровайна, Наталія Веселовська, Ганна Зайченко, Ніна Луценко, Олексій Путієнко, Андрій Сергієнко, Надія Ульянова, Валерій Беляєв, Олександра Павлів, Діана Чічур, Василь Шевчик

This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International (CC BY 4.0) that allows users to read, download, copy, distribute, print, search, or link to the full texts of the articles, or use them for any other lawful purpose, without asking prior permission from the publisher or the author as long as they cite the source.
COPYRIGHT NOTICE
Authors who publish in this journal agree to the following terms:
- Authors hold copyright immediately after publication of their works and retain publishing rights without any restrictions.
- The copyright commencement date complies the publication date of the issue, where the article is included in.
DEPOSIT POLICY
- Authors are permitted and encouraged to post their work online (e.g., in institutional repositories or on their website) during the editorial process, as it can lead to productive exchanges, as well as earlier and greater citation of published work.
- Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work with an acknowledgement of its initial publication in this journal.
- Post-print (post-refereeing manuscript version) and publisher's PDF-version self-archiving is allowed.
- Archiving the pre-print (pre-refereeing manuscript version) not allowed.